|
Executives who carry vast experience about FDA Regulated Industry and Experts get down to discussing industry-related best practices, regulatory updates, changes in technologies, and much more relating to FDA Regulated Industry.Īs a sponsor of these seminars, you get the opportunity to have your product and company reach out to C-Level executives in FDA Regulatory -related industries and become known among these elite executives and subject matter experts. Sponsorship Program benefits for “Quality Assurance Auditing for FDA Regulated Industry” seminarĪt this seminar, world-renowned FDA Regulated Industry subject matter experts interact with CXO’s of various designations. Until December 10, Early Bird Price: $1,295.00 From December 11 to January 23, Regular Price: $1,495.00 Price: $1,295.00 (Seminar Fee for One Delegate) Venue: Courtyard Arlington Crystal City/Reagan National Airport2899 Jefferson Davis Highway Arlington, VA 22202 USA Location: Washington, DC Date: January 25th & 26th, 2018 and Time: 9:00 AM to 6:00 PM He has a Masters Degree in Mathematics is an ASQ certified Biomedical Auditor, Quality Auditor, Quality Engineer, Reliability Engineer, and Six Sigma Black Belt and certified by APICS in Resource Management. Dan has more than 30 years' experience in quality, operations, and program management in regulated industries including aviation, defense, medical devices, and clinical labs. 
Lecture 4: Reporting to Management Reviewĭan O'Leary is President of Ombu Enterprises, LLC, a company offering training and execution in Operational Excellence, focused on analytic skills and a systems approach to operations management. Lecture 3: Responding to audit nonconformances Lecture 2: Being Audited (2nd and 3rd party) Lecture 1: Conducting Second Party Auditsĭocumenting the need for corrective action Qualifying auditors - Training and experienceĪssigning auditors - Avoiding conflicts of assignment Lecture 3: Auditor Qualifications and Assignmentĭetermine auditor competence requirements Lecture 1: Audit requirements in QMS standardsĭeveloping and maintaining the audit scheduleĭetermining audit areas - process, procedure, or department Understand the role of correction and corrective action related to an audit nonconformanceĭevelop an effective approach for reporting at Management Review Recognize which audits an auditor could conduct and which ones to avoid Recognize the role of auditor competency and how to determine it Understand audit scheduling and methods to establish and update the schedule Learn the difference among audit methods (system, product, process, etc.) Learn the difference among the audit types The workshop discusses the role of management review and helps identify the appropriate statistical methodologies for effective communication to management. The workshop presentation should use statistical methodology to analyze quality audit reports. Management needs to understand the results from each audit type as part of Management Review. The workshop provides methods and exercises to for these important activities. An effective audit program determines and eliminates the causes of the nonconformity, corrective action, to prevent recurrence. This includes correction, investigation, and corrective action. The workshop explains how to implement these quality audit program elements using examples and exercises that help develop the necessary skills.Īnother important part of an audit program is responding to audit nonconformances. Assign qualified people based on training, skill, and conflict of interest considerations. Develop a plan to schedule, conduct, report, and close audits. The program includes the two major elements. This workshop provides the tools to establish and maintain an effective quality audit program for medical device manufacturing. Independent audits (3rd party) can help provide assurance about the health of a QMS. Customer/supplier audits (2ndparty) play a role in an effective supplier management program your company could be the subject of the audit. Internal quality audits (1st party) help a company evaluate itself. In this workshop, participants examine the various types of audits to understand their role. Audits are one of the most powerful tools that management has to assure on effective QMS. Quality audits are an important element in implementing and maintaining an effective Quality Management System (QMS). 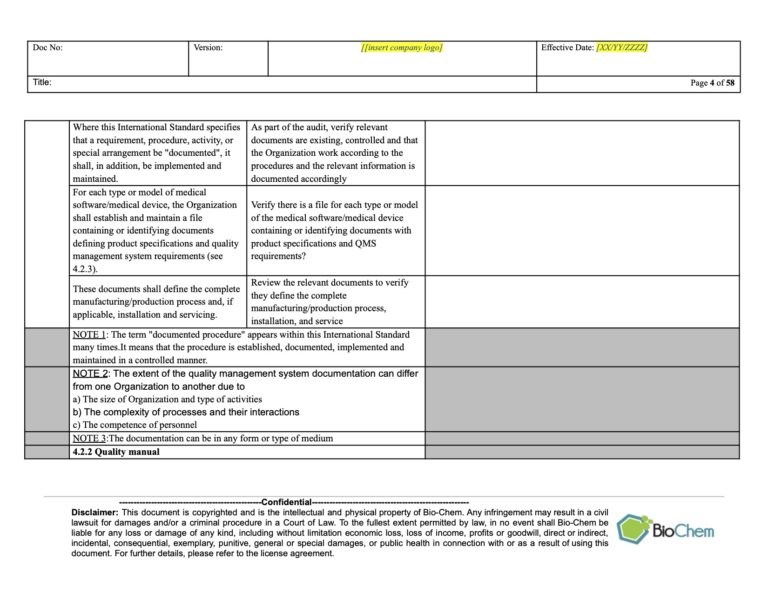
Course "Quality Audits for the Medical Device Industry" has been pre-approved by RAPS as eligible for up to 12 credits towards a participant's RAC recertification upon full completion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
